Testing tumors with next-generation sequencing in patients with ovarian cancer can provide teams insight into targetable mutations that may affect treatment decisions, although this approach is not the current standard of care for all patients, an expert said.
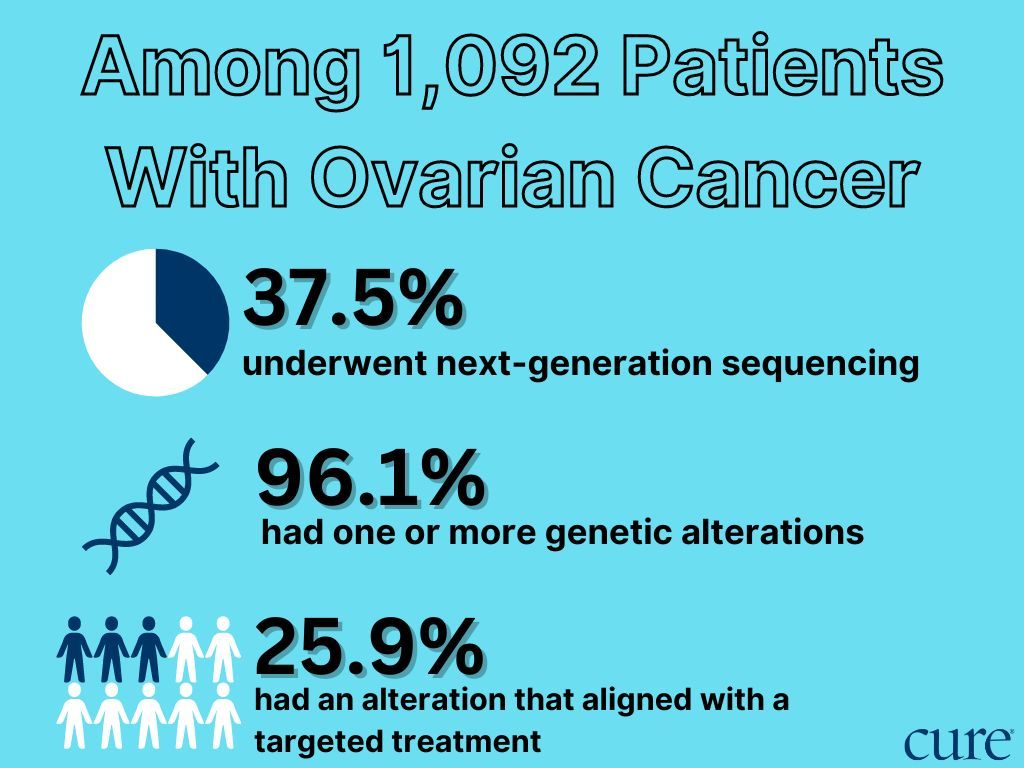
In an analysis of more than 1,000 patients with ovarian cancer, nearly all of them had a genetic alteration — and 25.9% were eligible for certain treatments based on those findings.
As more personalized medicines come into the space, having a better idea of the tumor’s genetic makeup will be instrumental, explained Dr. Anil K. Sood, professor and vice chair for translational research in the departments of gynecologic oncology and cancer biology at The University of Texas MD Anderson Cancer Center in Houston.
Sood and colleagues recently conducted a study that looked at patients with high-grade epithelial ovarian carcinoma and found that among patients who underwent next-generation sequencing — which is when the tumor is sampled and tested for mutations — 96.1% had at least one mutation, while 25.9% had a mutation that aligned with a specific targeted treatment that could work for their cancer.
“As science and clinical medicine continue to evolve, there are a lot of new technologies coming about,” Sood said in an interview with CURE®.
Why is next-generation sequencing in ovarian cancer an important topic to research?
Patients with ovarian cancer still, unfortunately, have high mortality rates. So we really want to do things to help improve or change the those outcomes.
Another aspect that’s very different now than perhaps even 10 or 11 years ago, is that the number of new drugs that are coming out is just incredible. A lot of the new drug development is very much focused on molecular or targeted pathways, rather than the old drugs we used to use related to chemotherapy and so on.
So there are a lot of technologies that could potentially help (determine) who may or may not be a candidate for certain types of drugs. We feel that that looking at sequencing of tumors where we can look at abnormalities such as genetic mutations within the tumor is such an assay where new drugs could potentially be paired depending on what those abnormalities show. That was a motivation (behind the study), to look at how often are we getting these assays, and then if we get these tests, are they really useful?
With all of these new drugs either approved or coming down the pipeline for ovarian cancer, is it current standard of care to get next-generation sequencing after an ovarian cancer diagnosis?
So right now, most (medical) societies certainly recommend germline testing, meaning testing for BRCA1- (and) BRCA2-related mutations using a blood test, be offered to all patients. That offering, obviously, has to be done in collaboration with genetic counselors. We’re finding as a part of our Cancer Moonshot program that if we do such testing for all women with ovarian cancer, especially those with high-grade serous, endometrioid, (or) those kinds of subtypes, that almost 18% of patients will have a germline mutation, which is quite a lot. There are already FDA-approved drugs such as PARP inhibitors that can be used (for patients with these mutations).
Now another exciting part, though, is that if the germline test is negative, then we’ll do what’s called reflex testing, looking at the tumor, or those kinds of BRCA1/2-related mutations, and also look for a test called HRD, or homologous recombination defect, because all of those relate to use of PARP inhibitors.
So regarding next-generation sequencing, is it approved for use for broader applications? Not yet, but, you know, at least the BRCA1/2 and related pathways, it certainly is very important for that purpose. And in that setting, the germline testing, historically, we would tell patients that it’s important for other family members in counseling and prevention and things like that for them. But at this point, it’s really important for the patient herself in terms of considering PARP inhibitor maintenance strategies, not just for the family members.
What can a patient expect when undergoing next-generation sequencing?
This kind of testing, the next-generation sequencing for broader panel of genes will typically (be done) if somebody develops relapsed cancer. And the reason that, at times, we’ll wait for that is that these panels continue to grow, as do the number of drugs coming out into clinical trials. And in that setting, I’d rather get the most current version of the test that looks at the broadest number of genes.
So (during) the process, I’ll basically talk to a patient directly (and say), “Listen, this kind of testing may be useful for figuring out possible clinical trials, possibly some of the new drugs in terms of matching towards your cancer type, and so on.” And then we’ll also get their permission to be able to send their tumor for that testing. Now, in addition to the tumor being tested, a blood sample is also important, because that serves as the normal control to know whether a genetic change in the tumor really is a mutation, or it’s just a variant in that individual that that exists. So sorting out that difference is important.
Since we’ve worked on this paper, another really exciting development occurred: a drug called an antibody drug conjugate, (Elahere), got FDA approved. Now, the test for that is not next-generation sequencing-based; we’re seeing a whole array of technologies coming out. And the test for that is more stain-based.
So now, we’re starting to test for that in patients who have relapsed disease to see if they might be candidate for that kind of a new drug.
This article was published by: Cure.com