Key takeaways:
- Researchers reported statistically significant improvement in median PFS with the combination added to standard of care in non-BRCA-mutated disease.
- The relative contribution of durvalumab requires further study.
CHICAGO — First-line chemotherapy plus bevacizumab and durvalumab — followed by maintenance durvalumab, bevacizumab and olaparib — extended PFS compared with standard of care for women with non-BRCA-mutated advanced ovarian cancer.
The findings of an interim analysis of the three-arm, randomized phase 3 DUO-O trial, presented at ASCO Annual Meeting, also noted a numerical improvement in PFS among patients who received chemotherapy, bevacizumab and durvalumab followed by maintenance bevacizumab and durvalumab, when compared with standard of care, but the difference did not reach statistical significance.
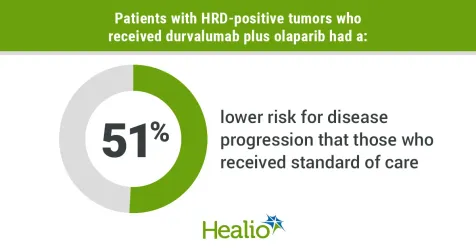
Data derived from Harter P, et al. Abstract LBA5506. Presented at: ASCO Annual Meeting 2023; June 2-6, 2023: Chicago.
“Maintenance therapy with olaparib, with or without the addition of bevacizumab, has improved outcomes in the first-line treatment of advanced ovarian cancer,” Carol Aghajanian, MD, medical oncologist at Memorial Sloan Kettering Cancer Center, said during a press conference. “However, there remains an unmet need … especially in some patient subgroups without a BRCA mutation.
“The relative contribution of durvalumab (in this study) requires further investigation,” she added.
Background and methodology
The DUO-O trial evaluated the efficacy and safety of paclitaxel and carboplatin with bevacizumab (Avastin, Genentech) and durvalumab (Imfinzi, AstraZeneca), followed by maintenance bevacizumab, durvalumab and olaparib (Lynparza; AstraZeneca, Merck) among women with advanced ovarian cancer and no BRCA1/BRCA2 mutations in the first-line setting.
Eligible patients had newly diagnosed stage III or stage IV, high-grade epithelial disease and had previously completed upfront or were to receive interval debulking surgery and one cycle of paclitaxel/carboplatin with or without bevacizumab.
Researchers randomly assigned the 1,130 patients to one of three groups. Group 1 (n = 378) received 15 mg/kg IV paclitaxel/carboplatin once every 3 weeks plus durvalumab or placebo for up to six cycles, followed by maintenance 15 mg/kg IV bevacizumab once every 6 weeks for a total of 15 months plus durvalumab or placebo and olaparib, a poly(ADP-ribose) polymerase (PARP) inhibitor, for 24 months.
Group 2 (n = 374) received paclitaxel/carboplatin plus bevacizumab and 1,120 mg IV durvalumab once every 3 weeks followed by maintenance bevacizumab and durvalumab and olaparib or placebo.
Group 3 (n = 378) received paclitaxel/carboplatin plus bevacizumab and durvalumab followed by maintenance bevacizumab, durvalumab and 300 mg twice-daily tablets olaparib.
PFS in group 3 vs. group 1 served as the primary endpoint. Researchers tested PFS first in patients with homologous recombination deficiency (HRD) and then the intent-to-treat population.
Results
Researchers observed a statistically significant and clinically meaningful improvement in PFS among patients in group 3 vs. group 1 in the HRD-positive population (37.3 months vs. 23 months; HR = 0.49; 95% CI, 0.34-0.69) and the intent-to-treat population (24.2 months vs. 19.3 months; HR = 0.63; 95% CI, 0.52-0.76).
Researchers also observed a consistent PFS effect in the HRD-negative subgroup (HR = 0.68; 95% CI, 0.54-0.86).
Data showed a numerical improvement in PFS in group 2 vs. group 1, but the difference did not achieve statistical significance by prespecified interim analysis.
Serious adverse events occurred in 34% of patients in group 1, 43% in group 2 and 39% in group 3.
With the trial ongoing, final PFS, OS and other key secondary results will be reported at a later date.
References:
- Addition of olaparib and durvalumab prolonged progression-free survival in patients with advanced ovarian cancer (ASCO press release). Published June 3, 2023. Accessed June 3, 2023.
- Harter P, et al. Abstract LBA5506. Presented at: ASCO Annual Meeting 2023; June 2-6, 2023: Chicago.
This article was published by: Helio


