By Andrea S. Blevins Primeau, PhD, MBA
A Ludwig Cancer Research study has shown that combining adoptive T cell therapy (ACT) with an innovative, personalized cancer vaccine under development at the Lausanne Branch of the Ludwig Institute for Cancer Research can benefit patients with late-stage, drug-resistant ovarian cancer.
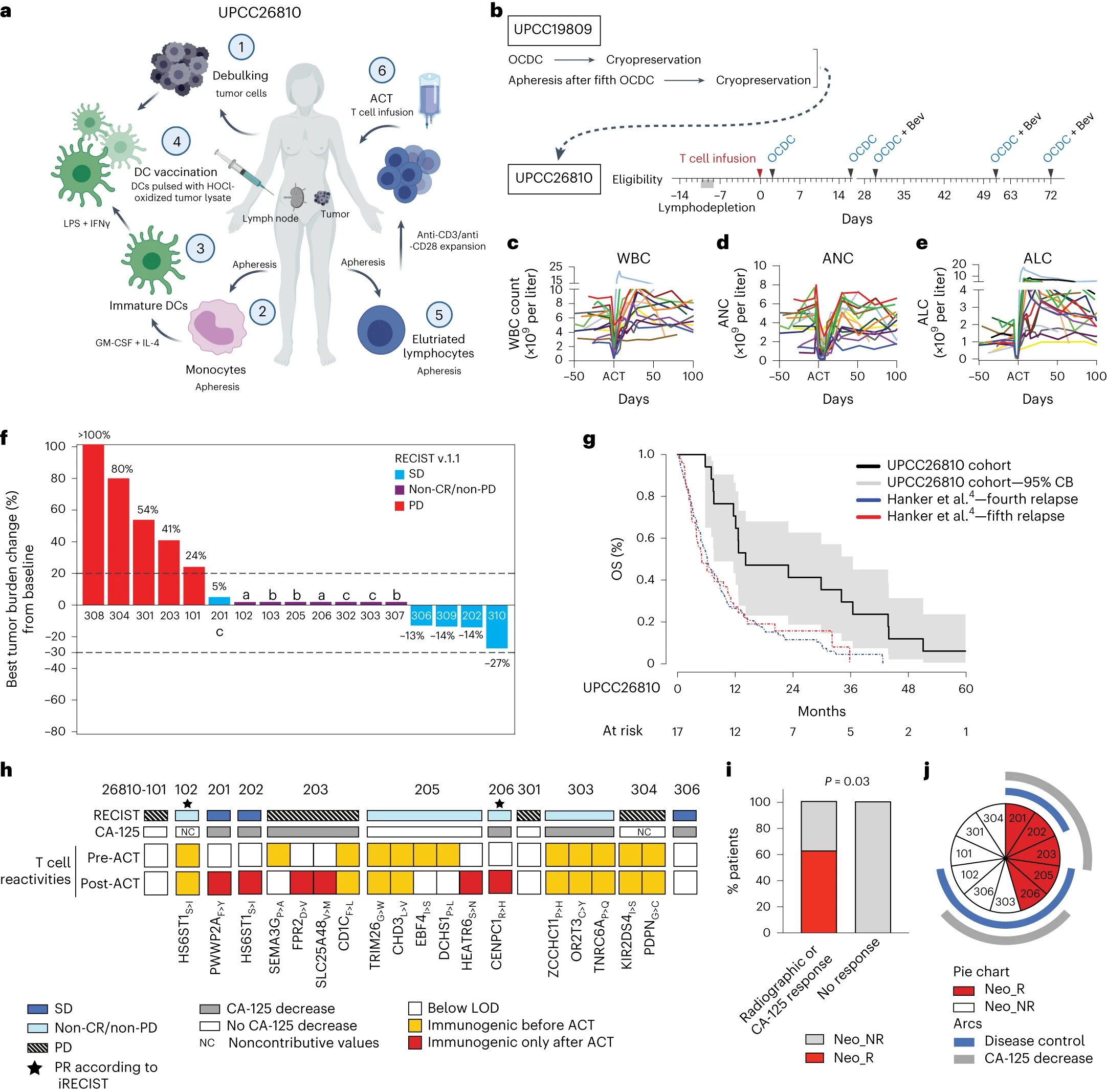
Researchers led by Ludwig Lausanne’s Sara Bobisse, Alexandre Harari, Lana Kandalaft and Director George Coukos analyzed responses to the combination therapy of 18 patients with advanced ovarian cancer who had previously participated in a clinical trial evaluating a therapeutic regimen that incorporated the personalized cancer vaccine.
In the current study—which, like the previous one, was done in partnership with researchers at the University of Pennsylvania—patients received an infusion of their own vaccine-primed, circulating immune cells (specifically, T cells), followed by multiple periodic doses of their personalized vaccines.
The researchers report today in Nature Cancer that combining the personalized cancer vaccine with ACT yielded control of the disease within three months in 12 of 17 patients. The treatment was also found to be safe and relatively well tolerated.
“This treatment could not eliminate tumors, but it did provide considerable benefit to many of the patients, who had all undergone extensive treatment prior to enrolling in the trial and were in the late stages of the disease,” said Kandalaft.
Though not a double-blind, placebo-controlled trial, the study’s findings suggest the benefits of the combination therapy were significant: the median overall survival time of patients who completed the regimen was 14.2 months, compared to a median historical survival of six months or less for comparable patients receiving fourth- and fifth-line chemotherapy.
“It was also exciting to be able to suggest, through sophisticated immunologic analysis, the mechanisms underlying treatment efficacy,” said Harari.
Ovarian cancer, like many other malignancies, has so far proved largely resistant to immunotherapies, most of which harness killer T cells, which destroy sick and infected cells. Ovarian cancer cells do, however, express neoantigens, which are randomly mutated proteins that can activate anti-tumor T cell responses.
Multiple approaches are today being developed at Ludwig Lausanne and elsewhere to target neoantigens for cancer immunotherapy. The personalized dendritic cell vaccine, developed over the past dozen years under the leadership of Kandalaft and Coukos, is one of them.
To make the vaccine, researchers first coax precursor immune cells isolated from patients to turn into dendritic cells, which present cancer antigens to killer T cells to direct and activate their anti-tumor responses.
These dendritic cells are then expanded in culture and pulsed with an extract of cancer cells from a patient’s tumor that is chemically treated to improve uptake and processing of antigens by the cultured immune cells. The vaccine is then injected into its corresponding patient’s lymph nodes, the anatomical site where dendritic cells activate anti-tumor T cells.
Patients enrolled in the trial received a therapeutic regimen that was shown to be promising in the previous clinical study. It involves delivering the dendritic cell vaccine along with bevacizumab, a standard ovarian cancer therapy that targets VEGF-A. Aside from promoting blood vessel formation, this factor, secreted by cancer cells, also hampers killer T cell entry into tumors.
Patients additionally receive low doses of chemotherapy to clear existing T cells from lymph nodes and make room for the newly infused T cells. A component of the chemotherapy, cyclophosphamide, also suppresses regulatory T cells, which inhibit killer T cell responses.
In the current study, patients received chemotherapy followed by ACT using vaccine-primed T cells that were expanded in culture in the presence of stimulatory immune factors. The ACT was followed by several cycles of vaccination.
“We were very happy that we could demonstrate how the combination therapy improved anti-tumor immune responses, and that those changes correlated to patient benefit,” said Bobisse. “Most notably, we could show that T cells targeting the neoantigens were reinvigorated by the combination therapy and correlated with positive responses to the therapy.”
Further, DNA sequences encoding neoantigens targeted by the T cells were found at higher levels in circulating tumor DNA, suggesting they had invited the destruction of tumor cells.
“This study illustrates how rational approaches to the design of immunotherapies can help overcome the barriers to immune responses that are erected by a variety of cancers, not least ovarian cancer,” said Coukos. “It has also taught us valuable lessons that we will be sure to apply to improve the efficacy of personalized cancer immunotherapies we’re developing here at Ludwig Lausanne.”
This article was published by: Medical Xpress


